Psychedelics
Oregon Completes First Full Year of Psilocybin Services Data Collection
SALEM – Oregon Psilocybin Services (OPS) has wrapped up its 2025 quarterly reporting, giving the public its clearest view yet of how the state's regulated program performed over a full calendar year. Psychedelic Alpha released its summary of the Q4 data, drawing from official state dashboards to highlight trends in client numbers, session formats, safety records, and who is actually using the services. Since OPS launched in January 2023, an estimated 16,000 clients have received psilocybin sessions, driving the sale of over 37,000 psilocybin products and generating more than $1.7 million in revenue. That demand has been consistent. What has changed is pricing. The average price per product has declined ...
Definium Doses First Patient in Second Phase 3 LSD-Based Trial
NEW YORK – Definium Therapeutics Inc. has taken another step forward in its late-stage testing of a psychedelic-derived treatment for major depressive disorder. The company announced that the first participant received a dose in the Ascend study, its second Phase 3 trial of DT120 ODT. DT120 ODT is Definium’s proprietary orally disintegrating tablet formulation of lysergide tartrate, a pharmaceutical version of LSD. The drug belongs to the class of serotonergic psychedelics and acts as a partial agonist at serotonin 2A receptors. It uses fast-dissolve technology aimed at providing faster absorption, better bioavailability, and fewer gastrointestinal side effects compared with traditional forms. The Ascend trial is a randomized, double-blind, placebo-controlled study ...
Congress Presses FDA on Psychedelic Therapy Approvals
WASHINGTON – A bipartisan coalition of 32 House members sent a formal letter to FDA Commissioner Martin A. Makary urging the agency to move faster on ongoing reviews of psychedelic treatments. The letter stated that "for many individuals, current treatment options remain insufficient, deepening an already urgent public health crisis," and referenced veterans, clinicians, and families as constituencies directly affected by the lack of approved alternatives. The congressional letter arrives weeks after a significant executive action from the White House. On April 18, 2026, President Trump signed an executive order directing federal agencies to accelerate development and potential approval of psychedelic-based therapies for conditions including PTSD, depression, and substance use ...
Psilocybin Shows Promise for Those Ready to Break Free from Nicotine
BALTIMORE – Researchers at Johns Hopkins University have published findings from a pilot randomized clinical trial that points to potential advantages of psilocybin-assisted therapy over standard nicotine replacement for helping adults quit cigarettes. Published in JAMA Network Open, the study compared nicotine patches to psilocybin as smoking cessation tools. At the six-month mark, participants who received just one dose of psilocybin had more than six times greater odds of remaining cigarette-free than those who relied on the nicotine substitute. The trial, led by Dr. Matthew Johnson, a professor of psychiatry at Johns Hopkins, enrolled 82 adult smokers who had a mean age of about 48, reported a median of six ...
Helus Pharma Teams Up with TARA Mind on Veteran Mental Health Efforts
NEW YORK – Helus Pharma, a clinical-stage biopharmaceutical company focused on developing novel serotonergic agonists (NSAs) to treat mental health conditions, announced it has joined forces with TARA Mind to help recruit veterans for its Phase 3 trial of an experimental treatment for major depressive disorder (MDD) and to broaden mental health support in military communities. The agreement brings together Helus Pharma’s PARADIGM HLP003 program and the networks of TARA Mind, which partners with Veterans Exploring Treatment Solutions (VETS). The groups plan to connect veterans with information about the ongoing trial and raise awareness of new research options for conditions like depression that affect service members at high rates. HLP003 ...
AtaiBeckley Reports Expanded Phase 2a Data for EMP-01 in Social Anxiety Disorder
NEW YORK – AtaiBeckley Inc., the clinical-stage biopharmaceutical firm born from the November merger of atai Life Sciences and Beckley Psytech, released additional results from its exploratory Phase 2a trial of EMP-01, an oral formulation of R-MDMA, in adults with moderate to severe social anxiety disorder (SAD). The study, conducted at seven sites in the United Kingdom, enrolled 71 participants who received two 225-milligram doses of the drug or placebo, administered 28 days apart in a clinic setting without any accompanying psychotherapy. The trial met its primary goal of demonstrating safety and tolerability, with no serious adverse events reported and most side effects described as mild to moderate and self-resolving. ...
President Trump Signs EO to Accelerate Psychedelic Research and Access
WASHINGTON – President Donald Trump signed an executive order directing federal agencies to speed up reviews of certain psychedelic compounds and open new pathways for patient access as treatments for serious mental health disorders like severe depression, PTSD, and addictions that resist standard therapies. The order, formally titled “Accelerating Medical Treatments for Serious Mental Illness,” instructs the Food and Drug Administration (FDA) to issue Commissioner’s National Priority Vouchers for psychedelic drugs that already hold Breakthrough Therapy designations. It also calls on the FDA and Drug Enforcement Administration (DEA) to create a process allowing eligible patients to obtain investigational psychedelics [with ibogaine compounds highlighted] under the Right to Try Act once ...
Compass Pathways Opens U.S. Grant Program for Psychedelic Therapy Training
NEW YORK – Compass Pathways PLC ADR announced a new grant program inviting U.S.-based organizations to develop training content for healthcare providers who could deliver its investigational COMP360 psilocybin treatment if regulators approve it. The London-based biotechnology company, which maintains offices in New York, said the grants will support the creation of structured materials focused on foundational psychedelic care plus a dedicated module on COMP360. The program builds on training already provided by established organizations that have prepared thousands of providers in psychedelic-assisted approaches. Compass plans to work directly with selected recipients to refine the content as it readies sites of care for a possible commercial launch of the therapy ...
9th Circuit Orders U.S. Government to Pay Over $2M to Ayahuasca Church
PHOENIX – The Ninth U.S. Circuit Court of Appeals has ruled that the federal government must reimburse the Church of the Eagle and the Condor and its members more than $2 million in attorney fees after a settlement that permits the group to import and use ayahuasca for religious ceremonies. The three-judge panel reversed a U.S. District Court decision in Arizona that had denied the fees, finding that the church qualified as the prevailing party under federal law once the government agreed to resolve the case. The unpublished memorandum opinion remanded the matter to District Judge Susan R. Bolton with instructions to award the fees and costs. The Church of ...
New Mexico Allocates $630K for Medical Psilocybin Treatment Equity Fund
SANTA FE – Gov. Michelle Lujan Grisham signed the state’s annual budget bill, setting aside $630,000 to create the Medical Psilocybin Treatment Equity Fund and extend access to supervised psilocybin therapy for low-income and rural New Mexicans. The allocation appears in House Bill 2 and backs a provision written into the New Mexico Medical Psilocybin Act, which the Legislature passed in 2025. That law established a regulated program allowing qualified patients to receive psilocybin-assisted therapy for treatment-resistant depression (TRD), post-traumatic stress disorder (PTSD), substance use disorders, and end-of-life distress. The equity fund, housed in the state treasury, will help cover treatment costs for patients who meet income criteria set by ...
Deutsche Bank AG Initiates Coverage of AtaiBeckley
NEW YORK – Deutsche Bank AG has begun formal research coverage of AtaiBeckley Inc., the Nasdaq-listed biopharmaceutical company created from the merger of atai Life Sciences and Beckley Psytech. David Hoang, a senior equity research analyst at Deutsche Bank who focuses on publicly traded companies in the healthcare and biotech sectors, assigned a ‘Buy’ rating and a $12 price target to the AtaiBeckley stock. The research reflects the bank’s evaluation of the company's portfolio of investigational treatments for mental health conditions often resistant to standard therapies. AtaiBeckley’s programs include BPL-003, a nasal spray formulation of mebufotenin (5-MeO-DMT) for treatment-resistant depression (TRD), and VLS-01, a DMT-based buccal film designed for the ...
Louisiana Bill Would Tap Opioid Settlement Funds for Psychedelic Therapy Studies
BATON ROUGE – Louisiana lawmakers have advanced legislation that would let the state direct money from opioid lawsuit settlements toward clinical research on psychedelic-assisted therapy. Senate Bill 43, sponsored by Sen. Patrick McMath (R), would create the Psychedelic-Assisted Therapy Program inside the Louisiana Department of Health. The measure targets studies on ibogaine and psilocybin [and related compounds] for people dealing with opioid use disorder, other substance use issues that occur alongside it, and certain treatment-resistant mental health or neurological conditions. Under the bill, academic health centers would run the trials in controlled hospital or clinic settings. Researchers would need full federal approvals, including an investigational new drug application from the ...
Gilgamesh Pharma Raises $60M to Fund Its Remaining Neuropsychiatric Assets
NEW YORK – Gilgamesh Pharma, the clinical-stage neuroscience company formed last year after AbbVie acquired its predecessor’s lead psychedelic-derived program, announced it had completed an oversubscribed $60 million Series A financing. The round was led by Satori Neuro and included participation from Prime Movers Lab, which backed earlier rounds of the original Gilgamesh Pharmaceuticals, along with other new and returning institutional investors. The proceeds will advance the company’s pipeline of proprietary neuropsychiatric candidates and expand its discovery platform. All programs are new chemical entities protected by composition-of-matter patents. Gilgamesh Pharma inherited its current assets from the 2025 AbbVie transaction, which acquired the $1.2 billion bretisilocin program [a short-acting 5-HT2A agonist ...
AbbVie Secures First Psychedelic PRIME Designation in Europe
BRUSSELS – The European Medicines Agency (EMA) has granted Priority Medicines (PRIME) eligibility to bretisilocin, an investigational compound from AbbVie Inc. aimed at treating major depressive disorder (MDD). The decision adopted at the Committee for Medicinal Products for Human Use meeting positions the drug as the first psychedelic candidate to receive this designation. PRIME offers early and ongoing dialogue with regulators to streamline development for medicines that address unmet needs. It functions much like the FDA’s Breakthrough Therapy Designation but stops short of any approval guarantee. Psychiatry has seen limited use of the program compared with fields like oncology, where dozens of candidates have qualified over the years. Bretisilocin, a ...
Massachusetts Lawmakers Push Forward Psychedelic Therapy Programs
BOSTON – Massachusetts lawmakers have advanced legislation that would set up pilot programs for regulated psychedelic therapy, with one measure clearing a key committee in September 2025 and related proposals still under review. The Senate Bill S.1400, sponsored by Sen. Cindy Friedman, won favorable approval from the Joint Committee on Mental Health, Substance Use and Recovery. It directs the Department of Public Health to license up to three specialized mental health clinics for supervised administration of psychedelic materials to treat conditions like depression, anxiety, PTSD, and substance use disorders. Clinics would operate under strict rules set by the department, including on-site delivery by multidisciplinary teams, patient screening and ongoing data ...
Hawaii Moves Forward on Psychedelic Therapy Research Framework
HONOLULU – The Hawaii Senate approved legislation that would set up a state task force to examine ways to bring breakthrough mental health treatments, like psilocybin- and MDMA-assisted therapies, into clinical and policy discussions. Senate Bill 3199, sponsored by Sen. Chris Lee (D), cleared the chamber on a unanimous 24-0 vote with one senator excused. The measure now heads to the House of Representatives, where it received first reading on March 10 and was referred to the committees on Health and Human Services and Finance. The bill establishes the Mental Health Emerging Therapies Task Force, which would operate for two years under an entity experienced in scientific research and medical ...
Minnesota House Panel Passes Bill for Psilocybin Therapy Pilot
SAINT PAUL – A Minnesota House committee has cleared a measure to set up a limited program for psilocybin-assisted therapy, marking a significant step toward regulated access for adults facing certain mental health issues. Yesterday, the Health Finance & Policy Committee approved HF 2906 on a bipartisan voice vote, sending it next to the Commerce Finance & Policy Committee for review. Moderate partisan bill (Democrat 6-1) would reschedule psilocybin from Schedule I to Schedule IV in state law, acknowledging its potential medical value and lower risk profile. It creates a three-year pilot allowing up to 1,000 patients aged 21-plus with qualifying conditions like depression, PTSD, anxiety, substance use disorders, or ...
AtaiBeckley Reports Q4 and FY 2025 Financial Results
NEW YORK – AtaiBeckley Inc. disclosed its fourth-quarter and full-year 2025 financial results, showing a significant net loss driven by costs from its recent merger while highlighting progress in its clinical programs for depression and anxiety disorders. The company reported a full-year net loss of $660 million compared with $149.3 million in 2024. Much of the increase stemmed from a $530 million non-cash charge tied to acquiring in-process research and development during the November 2025 combination of atai Life Sciences and Beckley Psytech, which also involved a U.S. redomiciliation completed in December. Revenue climbed to $4.1 million for the year from $308,000 previously, reflecting modest growth in licensing and collaboration ...
Connecticut Lawmakers Expand Psychedelics Access
HARTFORD – Connecticut's Joint Committee on Public Health approved a bill that broadens the state's pilot program for psychedelic-assisted therapy, opening it to adults beyond just veterans and first responders. The measure, Senate Bill 191, clears the way for qualified patients to access MDMA or psilocybin treatments through FDA-approved research studies administered by a state medical school. The legislation strikes a prior requirement that the program end once the federal Drug Enforcement Administration approves MDMA and psilocybin for medical use. Instead, it mandates the Department of Mental Health and Addiction Services to run the initiative within available funds, focusing on clinical trials for those 18-plus who meet eligibility standards set ...
Psychedelic Alpha Released Q1 2026 Psychedelic Drug Development Bullseye Charts
LOS ANGELES – Psychedelic Alpha, the data-focused research and analysis group that monitors the psychedelics sector, published its latest Bullseye Charts showing the state of drug development programs as of the first quarter of 2026. The charts present the pipeline in two formats. One displays the full spectrum of candidates from discovery through potential approval. The second focuses on assets in Phase 2 and later stages, grouped by broad therapeutic indications like depression, anxiety, post-traumatic stress disorder, and substance use disorders. Concentric rings mark each development stage, with the center reserved for approved products. The update reflects several corporate name changes that occurred in late 2025 and early 2026. MindMed ...
Single DMT Dose Shows Promise in Cutting TRD Symptoms
NEW YORK – A single intravenous dose of the psychedelic compound dimethyltryptamine (DMT), combined with psychological support, produced rapid and sustained reductions in depressive symptoms among adults with moderate-to-severe major depressive disorder, according to results from a small Phase 2a clinical trial. The study, published in Nature Medicine, involved 34 participants with treatment-resistant depression (TRD). Researchers from Imperial College London and collaborators conducted a double-blind, placebo-controlled trial in which participants received either a 21.5 mg dose of DMT fumarate (SPL026 by Helus Pharma Inc.) or placebo infused over 10 minutes. All received preparatory and integrative psychotherapy sessions. The primary endpoint was the change in Montgomery–Åsberg Depression Rating Scale (MADRS) scores ...
Compass Pathways Advances in Second Phase 3 Trial for Psilocybin-Assisted TRD
LONDON – Compass Pathways PLC ADR, a UK-based mental health biotech company, announced that its second Phase 3 trial met the primary endpoint in evaluating COMP360, a synthetic formulation of psilocybin, for treatment-resistant depression (TRD). The COMP006 study, which involved 581 participants across North America and Europe, tested two fixed 25 mg doses administered three weeks apart against a 1 mg control dose. At the six-week mark, the 25 mg group showed a mean difference of -3.8 points on the Montgomery-Åsberg Depression Rating Scale (MADRS) compared with the control arm, with a p-value less than 0.001 and a 95% confidence interval of [-5.8, -1.8]. Thirty-nine percent of participants in the ...
New Hampshire House Advances Bill to Regulate Medical Psilocybin Use
CONCORD – The New Hampshire House of Representatives has given initial approval to a bipartisan measure that would establish a state program allowing supervised therapeutic access to psilocybin for qualifying medical conditions. House Bill 1809, sponsored by Rep. Buzz Scherr (D), cleared the chamber on a voice vote through its consent calendar. The legislation previously received unanimous support from the House Health, Human Services and Elderly Affairs Committee in a 18-0 vote late last month. The bill directs the Department of Health and Human Services (DHHS) to develop rules for a program providing psilocybin in controlled, medically supervised settings. It creates an advisory board to review clinical data, monitor patient ...
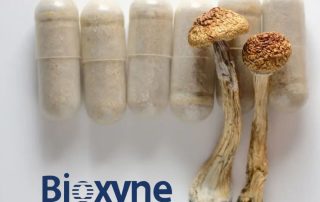
Bioxyne Lands Initial Orders for GMP-Compliant Psilocybin Capsules
SYDNEY – Bioxyne Limited, an Australian life sciences and wellness products company, has received its first commercial orders for GMP-manufactured psilocybin capsules through its subsidiary Breathe Life Sciences (BLS). The capsules, branded BLSPSIL25, are intended for investigational use in addressing treatment-resistant depression, as well as exploratory research into other mental health conditions like anxiety and substance use disorders, and PTSD. The initial orders cover 250 doses, sufficient to support treatment for about 60 patients over the next 12 months. Supplies are going to authorized prescribers in Queensland and Western Australia under Australia's Therapeutic Goods Administration (TGA) Authorised Prescriber pathway. This follows the company's 2024 achievement as the first [and for ...
Clearmind Featured in Bipartisan Veterans Therapy Bill
WASHINGTON D.C. – The recent introduction of bipartisan legislation in the U.S. House of Representatives has drawn attention to Clearmind Medicine Inc.'s proprietary compound MEAI as part of efforts to broaden veterans' access to certain emerging therapies. Clearmind announced that MEAI was explicitly named in H.R. 7091, the Expanding Veterans’ Access to Emerging Treatments Act of 2026. The bill directs the Department of Veterans Affairs to set up programs for investigational research and extended access to innovative treatments addressing unmet needs among veterans, particularly for conditions such as alcohol use disorder and PTSD. Alongside MEAI, the legislation lists psilocybin, MDMA, 5-MeO-DMT, ibogaine, ketamine, and other compounds that the VA secretary ...